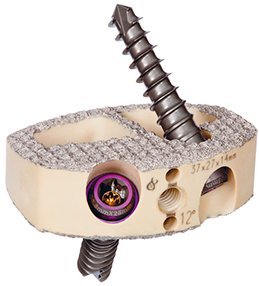
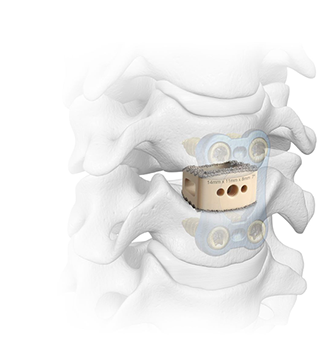
Formulated to Enhance Bone Fusion
- Viable spine-derived cells are collected from the vertebral body region of the donor and preserved with the use of a next generation DMSO-free cryoprotectant
- A demineralized bone scaffold with osteoinductive potential which provides exposure of signaling molecules and bone morphogenetic proteins1
DMSO-Free without compromise
- ≥150,000 cells / cc for osteogenic supplementation2
- Proprietary DMSO-free cryoprotectant that requires no rinsing or decanting steps required
- OR ease of use with no rinsing or decanting steps required
- 4-hour working window for implantation
Evidenced-Based Application: Fusion
- MIS-TLIF study demonstrated 96% fusion at 12 months3
- MIS-LLIF study demonstrated 94% fusion at 12 months4
1. Gruskin, E. et.al., Demineralized bone matrix in bone repair: history and use. Advanced Drug Delivery Reviews, 2012. 64:1063-1077.
2. Data on file at VIVEX Biologics, Inc.
3. Tally, William C, et al., Transforaminal Lumbar Interbody Fusion with Viable Allograft: 75 Consecutive Cases at 12-Month Follow-Up. Int J Spine Surg, 2018. Vol. 12, No. 1 p. 76-84.
4. Talley WC, Temple HT, Burkus JK Lateral lumbar intebody fusion using a cellular allogenic bone matrix in the treatment of symptomatic degenerative lumbar disc disease and lumbar spinal instability. Int J Spine Surg. 2021; 7(3):310-317