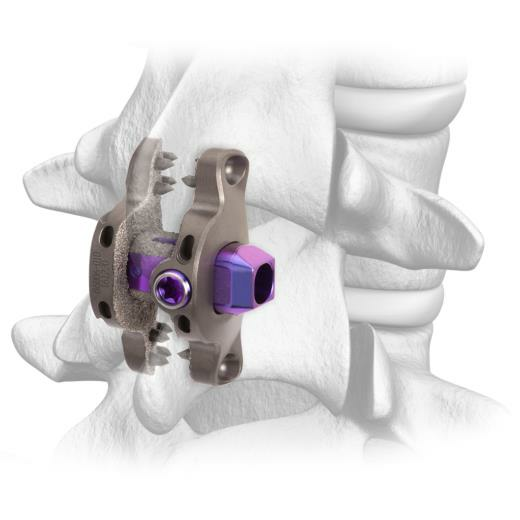
Spinal Elements’ Clutch is the only interspinous process device designed to maintain active compression against the spinous processes to resist loosening and migration.
Carlsbad, CA – May 24, 2018 – Spinal Elements, a spine technology company, today announced the release of its Clutch interspinous process device. This new product further enhances the breadth of Spinal Elements’ thoracolumbar portfolio and offers surgeons more options for treatment of various posterior thoracolumbar pathologies.
Clutch has been in use since shortly after its FDA 510(k) clearance in 2017 and with this expanded commercial release, Spinal Elements again sets the new standard with a unique and clinically relevant feature: The Clutch interspinous process device’s proprietary spring-activated mechanism is designed to maintain active compression against the spinous processes to resist loosening and migration. Additionally, the device’s bone opposing surfaces are enhanced with clinically proven Ti-Bond® technology, designed to provide immediate stability and long-term fixation.
“I have found interspinous process devices to be a very useful surgical option in my practice. Clutch has an ideal anatomic design that lets me confidently place the device in a more anterior position, fixating securely into the best quality bone”, said Dr. Burak Ozgur, a nationally recognized neurosurgeon in Newport Beach, California. Jason Blain, President and CEO of Spinal Elements added, “Creating procedural solutions for improved patient outcomes is a critical requirement for every system we design and market. Clutch meets this need with what we believe to be a best-in-class design. I am excited for how this addition complements and expands our product portfolio”.
About Spinal Elements
Spinal Elements is an outcomes-driven spinal surgical solutions company with locations in Carlsbad, CA and Marietta, GA. A leading designer, developer, manufacturer and marketer of innovative medical devices used in spinal surgical procedures, our mission is to combine leading medical device technologies, biologics and instrumentation to create positive surgical outcomes that exceed surgeon and patient expectations. For more information, please visit www.spinalelements.com.
For interviews or more information, contact:
Laura Charlton (formerly Johnson) for Spinal Elements laurajohnsonpr@yahoo.com (760) 450 – 7749 cell
PRESS RELEASE CL PR 00 ENO 220518